 Mariane Hodgkinson, hygiene specialist at Hillbrush looks at the challenges of implementing PRP programmes in food and beverage manufacture.
Mariane Hodgkinson, hygiene specialist at Hillbrush looks at the challenges of implementing PRP programmes in food and beverage manufacture.
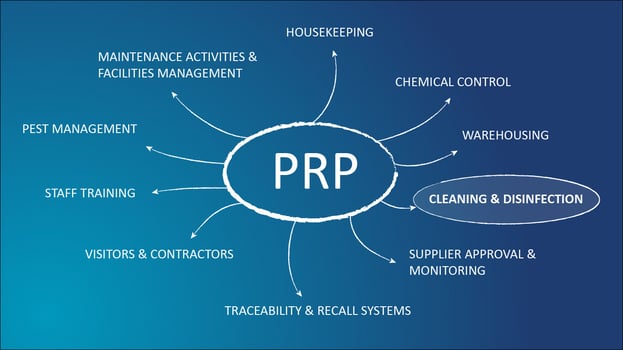
So many fundamental activities form the pre-requisite programmes (PRP) of food and drink manufacturing, with most of them part of Good Manufacturing Practices used throughout mass manufacturing. The usual suspects you may find within your PRP are pest management, staff training, visitors and contractors, traceability and recall systems, warehousing, cleaning and disinfection, supplier approval and monitoring, chemical control, housekeeping, maintenance activities and facilities management.
Most of these will be carried out by accredited specialist providers which should give you peace of mind that everything will be carried out according to best practice. It is easy to assume that once everything is in place with cleaning for example, that not much can go wrong. But sadly, this isn’t always the case.
There is no doubt that setting up cleaning protocols can be a tough job, involving many experienced personnel around the table discussing the needs of their specific areas, systems feasibility and implementation based on existing staff and resources, educating all relevant parties, setting up documented controls and monitoring records. But it is important that the work doesn’t stop there.
There will always be the need for continuous review of the PRP’s effectiveness and suitability, based on many different criteria including:
- new and existing product development
- plant/factory development
- new equipment and technology additions
- personnel change
- ageing of equipment
- changes happening inside and outside the factory
- changes in regulations
There isn’t a magic formula to consistently ensure full compliance to legal responsibilities, accreditations and internal standards. The starting point is to confirm that top level commitment and support is in place to ensure new requirements are being transferred to all levels of the business and are implemented effectively. Management has its part to play in monitoring activities, hazards and risks, relaying changes to operators in a clear and constructive way, and ensuring monitoring tools are being effective in identifying potential trends or areas of concern. Operators also have a vital part to play, as the food safety application will be in their hands as part of day-to-day activities. By understanding the reasons behind their work and how they affect the bigger picture, it will provide operators with the power to influence how well activities are being carried out and how they can be improved.
We can use cleaning and disinfection, as an example of an activity that will take place in all areas of the business, from keeping the perimeter tidy to ensuring the gasket inside the filler of a packing machine is appropriately clean and disinfected when starting a new production, ensuring product safety is under control. This activity has many levels of compliance, depending on the assessment of risk present in the specific manufacturing process or activity taking place.
Whilst perceived as a basic activity, complex training and resources are required during the set up and consequent changes. When we look at controls around cleaning and disinfection, we can start with the selection of who will be carrying out the work.
"Using internal personnel is my preferred option as the site has a greater opportunity to select individuals with the correct behaviour and personal characteristics that are beneficial to the activity"
e.g. methodical, attention to detail, good personal hygiene and food safety minded.
The other option is to use external providers, experts whose jobs are to ensure areas are cleaned to the agreed standards, using correct tools and methods. Using qualified professionals allows the site to focus on what they know best and ensure this key activity is being carried out by competent and fully trained people, with targets and whose full focus is to ensure the area is free from contamination. The issue here is that sometimes the site “washes its hands” of the activity when, in reality, this solution will also need to be accompanied by monitoring of results by the site, validation of cleaning and verification that activities are still being carried out as agreed, just as one would carry out for internal cleaning activities. Many sites will use a combination of both depending on the hazard present in the area and if related to any product contact areas.
Training in cleaning and disinfecting as well as chemical use can be a tricky aspect. The best approach is to ensure the training material is relevant, with practical aspects of the job and provides enough information so the workers can understand the hazards they are trying to control.
Different types of cleaning will require different tools, from fully automated systems to manual set-ups using handheld tools and anything in between. A lot of effort can be given to choosing the correct tools for the job, but unfortunately this can become less significant as systems become more established, even when changes take place. This, in turn, can give a false sense of security and compliance could have a knock-on effect on product safety. Review of the tools in use must be part of the hazard assessment, especially when new processes and products are being introduced. Sometimes even an add-on to processing equipment can change how you need to carry out the clean!
Cleaning and housekeeping go hand-in-hand, and improvement in one will very likely reflect in the other activity.
Documentation of activities and procedures are critical to ensure you have standards documented, information equally shared between all using similar language and evidence that this has been followed as intended. Improvements in processing lines may require changes in record completion, most certainly changes in procedures and a much-needed refresher in cleaning training, ensuring the importance of cleaning is brought back to everyone’s attention.
Many more aspects can be discussed about pre-requisites and the surrounding systems, but the aspects I have discussed above can be very easily transferred to other activities such as warehousing, facilities management, pest management to ensure food safety is being kept alive in your business.
If you need any further support, please contact Hillbrush.